|
Quality control material is material that is analyzed along with patient specimens and should be treated the same as patient specimens. Within the laboratory, one method that is used to ensure the quality of the patient results is to use quality control material. One technique that laboratories can use to check the quality assurance procedures is to follow random samples as they proceed from collection to analysis to charting of the results. Similarly, if a diluted sample’s result was not multiplied by the dilution factor correctly, that is a postanalytical error. If the sample is analyzed incorrectly, that is an analytical error. For example, if the wrong patient’s sample is drawn, that is a preanalytical error.  
The laboratory must monitor each of these activity phases for errors to ensure accurate results. Activities that occur before the sample reaches the laboratory are called preanalytical activities, those that occur in the laboratory that directly deal with the analysis of the sample are called analytical activities and those activities after the analysis is performed are called postanalytical activities. 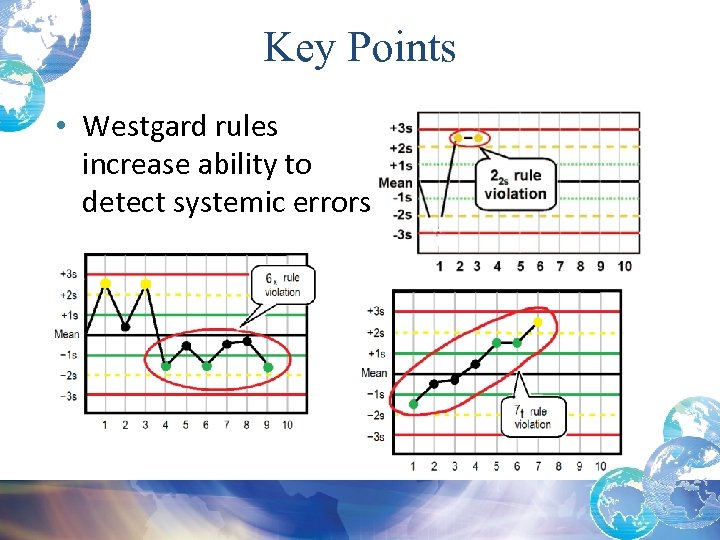 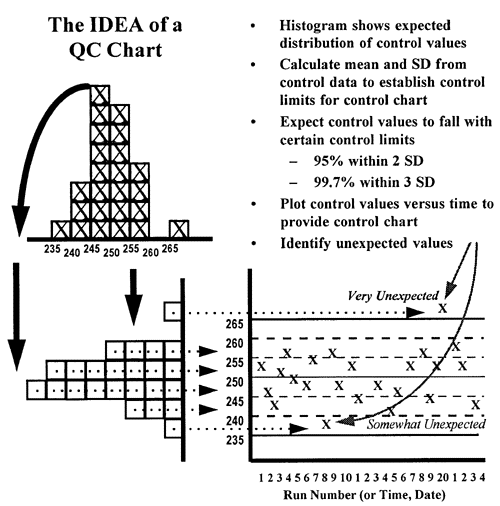
Quality assurance is the monitoring of any activity that is associated with a laboratory result. “Quality assurance” is the process in which this occurs. In the laboratory, the instruments and methodologies must be monitored to ensure accurate results.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
